 As a provider of stem cell products and technologies, you need to make effective product development decisions, generate improved revenues, and take market share from your competition. To do this, you need to be educated about prevailing market conditions. This involves knowing which stem cell types are showing the most promise and understanding methods through which they could be commercialized.
As a provider of stem cell products and technologies, you need to make effective product development decisions, generate improved revenues, and take market share from your competition. To do this, you need to be educated about prevailing market conditions. This involves knowing which stem cell types are showing the most promise and understanding methods through which they could be commercialized.
Stem cells are still a relatively new discovery, as the first stem cells were discovered in human cord blood in 1978, the first mouse embryonic stem cells were derived in 1981, and it was not until 2006 that induced pluripotent stem cells (iPSCs) were produced for the first time.
Induced Pluripotent Stem Cells
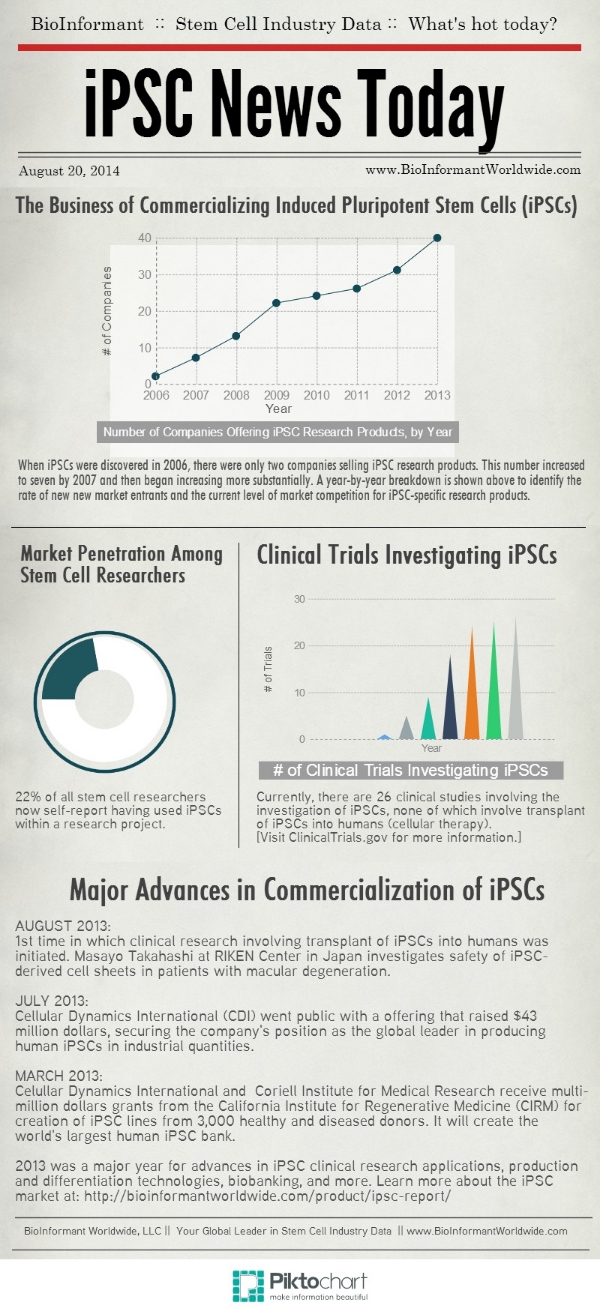
Since the discovery of iPSCs, a large and thriving research product market has grown into existence, largely because the cells are completely non-controversial and can be generated directly from adult cells. Today, the number of iPSC products sold worldwide is growing at an annual rate of 14.7%. In addition, 22% of all stem cell researchers now self-report having used iPSCs within a research project.
It is clear that iPSCs represent a lucrative product market, but methods for commercializing this cell type are still being explored, as clinical studies investigating iPSCs continue to increase in number. At this time, nearly all clinical studies involving iPSCs are for the creation and evaluation of iPSC lines from specific patient populations, in order to determine if these cell lines could be a good model for a disease of interest in that patient population. (See ClinicalTrials.gov for a current list of these trials.)
However, the first clinical study involving transplant of iPSCs into humans began in August 2013 to investigate the use of iPSCs in treating patients with macular degeneration. This clinical study is now underway at the Riken Center in Japan, led by Masayo Takahashi.
In addition, a new patent challenge has exploded onto the stem cell scene. The famous “Yamanaka Patent” (U.S. Patent No. 8,058,065) for cellular reprogramming of iPSCs is now being challenged in a patent lawsuit by a group called “BioGatekeeper.” If successful in canceling the Yamanaka Patent, this could create incredible innovation in the stem cell/regenerative medicine sector.
Due to these exciting and rapidly changing market conditions, the infographic below explores the business of commercializing iPSCs, including the production of research products, the funding of clinical research trials, and major advances in cellular therapy and biobanking.
We welcome your comments below and encourage you to claim guidance that will allow for your competitive advantage within the stem cell marketplace by viewing BioInformant's full report: “Complete 2013-14 Induced Pluripotent Stem Cell Industry Report.”
Thanks for reading!
Editor's Note:
This post was written by Cade Hildreth, CEO of BioInformant Worldwide, LLC.
About BioInformant:
BioInformant Worldwide, LLC, specializes in stem cell market intelligence. As a specialty research company, we use technology to track and identify profitable opportunities within the stem cell sector and provide this data to clients pursuing aggressive growth. We are the only market intelligence company that has specifically served the stem cell sector since it emerged.
To date, BioInformant has been featured on prominent news outlets that include Nature Biotechnology, the Wall Street Journal, CBS News, Yahoo Finance, Medical Ethics, MarketWatch, the Center for BioNetworking, and many more. Serving Fortune 500 leaders that include GE Healthcare, Pfizer, Goldman Sachs, Beckton Dickinson, Thermo Fisher Scientific, and many more, BioInformant is your trusted source of stem cell industry data.
BioInformant is also available on Profound.com...
Purchase individual report sections for a pro-rated price. Visit Profound.com to learn more.


