 Many have questioned whether China can move from "the world's factory” for consumer goods, electronics, and commodities, into technologically complex biologics.
Many have questioned whether China can move from "the world's factory” for consumer goods, electronics, and commodities, into technologically complex biologics.
However, the more relevant question is whether China can afford not to advance its healthcare segment. China is:
- A fifth of the world’s population
- Upwardly mobile, with a middle-class that will comprise 30% of the country’s population in 10 years. (Incidentally, that 30% is larger than the entire population of the US.)
- Set to eclipse the U.S as the largest retail market, growing at 7.5% to $5.6 trillion.
- In need of domestic biologics, as increasing affluence heightens demand and inevitably accelerates Chinese biomanufacturing growth
Considering these factors, China’s biologics industry growth seems like a foregone conclusion. So the key issue for the global biologics industry is this: How will the rest of the developed world’s biologics manufacturers, suppliers, distributors, and regulators participate?
The Chinese Biologics Market: 11 Key Data Points
This is an opportunity for the developed world’s biologics manufacturers, suppliers, distributors, and regulators to take advantage of the high-growth potential of China's biologics industry. As domestic growth increasingly becomes an engine for its global biopharma emergence over the next 10 years, we point to just a few key data points from BioPlan's peer-reviewed study Advances in Biopharmaceutical Technology in China, 2nd Ed.
- In 2018, the Chinese biopharmaceutical market reached over $9 billion, of which the domestic mAb market comprised a large percentage.
- Between 2010 and 2013, China’s biologics market grew 21% annually.
- The country is home to 180 biopharmaceutical manufacturing facilities with >850,000 L total capacity – larger than that of India.
- Of the 27 mAbs approved in China, 18 are imported and 9 are locally manufactured.
- China filed >1 million patents in 2015, almost double the number filed by U.S. inventors.
- Basic medical insurance and cooperative medical systems cover essentially all in China.
- In the past decade, total healthcare expenditures increased from around $150 billion to $4.8 trillion, growing from 4.5% to 6.2% of national GDP.
- At $122.6 billion, China was the world's 2nd-largest market for pharmaceuticals in 2017 (including small molecules).
- The Chinese pharmaceutical market has a CAGR of >15% (versus < 4% in developed countries).
- By 2020, the number of people over the age of 65 years in China will reach 170 million.
- 30% of the >1 million biomedical R&D staff in China are returnees.
China: A Top Outsourcing Destination for U.S. Biopharma Companies?
Just 7 or 8 years ago, few – if any – Western biologics manufacturers considered China a viable option for outsourcing production or services. Intellectual property issues, lack of awareness, and expectations of poor quality were the norm. That’s shifted dramatically, according to BioPlan's 15th Annual Report and Survey of Biopharmaceutical Manufacturing.
This past survey, BioPlan found that 25.4% of U.S. biopharma decision-makers now consider China to be a likely destination for outsourcing within the next in 5 years, with over 50% counting it as a possible destination.
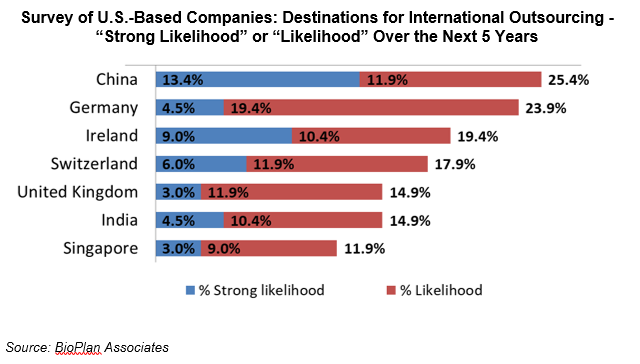
So although China biopharma at this point is mainly domestically directed, our research indicates that biomanufacturers in both China and in Western markets see a future where China is a viable partner, competitor, or supplier.
Interested in Learning More?
For a deeper dive into the Chinese biologics market, be sure to download Advances in Biopharmaceutical Technology in China, 2nd Ed, a co-publication between BioPlan and Society for Industrial Microbiology & Biotechnology. The report provides an authoritative analysis of the biopharmaceutical industry, as well as the state of China's technology.
Click the button below to view the full report page.
About the Author: Eric Langer is a Managing Partner at BioPlan, a biotechnology and healthcare marketing research and strategic consulting firm, providing services since 1989.


