 The laparoscopy market in the US has experienced significant growth due to the rising demand for minimally invasive surgical techniques. Laparoscopy involves small incisions which offer numerous benefits over traditional open surgery, such as reduced pain, shorter recovery times and smaller scars.
The laparoscopy market in the US has experienced significant growth due to the rising demand for minimally invasive surgical techniques. Laparoscopy involves small incisions which offer numerous benefits over traditional open surgery, such as reduced pain, shorter recovery times and smaller scars.
The market is driven by an increasing prevalence of chronic diseases and health issues, such as obesity, all of which have led to a rise in demand for minimally invasive surgeries. However, market growth is hindered by the high cost of laparoscopic procedures and equipment, as well as the negative impact of the COVID-19 pandemic on elective surgeries.
The US laparoscopy market is segmented by application into gynecological surgery, urological surgery, general surgery and other procedures. Gynecological surgery holds the largest market share due to the rising incidence of gynecological disorders and the increasing demand for minimally invasive hysterectomies. Technological advancements in laparoscopic equipment and techniques, including robotic-assisted laparoscopic surgery, provide greater precision and control during complex procedures. However, the adoption of robotic surgical platforms may cannibalize the traditional laparoscopic device market.
Some interesting trends affecting the market include the legislation of smoke evacuation laws, FDA warnings and the preference of using disposable devices due to heightened sanitary concerns since the COVID-19 pandemic. As the demand for minimally invasive surgical techniques continues to rise, the market is expected to see continued growth in the coming years. Nonetheless, it is important to address challenges like high costs and negative impacts of COVID-19 to optimize the potential of the laparoscopy market (iData, 2023).
Surgical Smoke Evacuation Laws
The implementation of surgical smoke evacuation laws in the United States has brought about significant changes in the laparoscopic insufflator market. These laws mandate the use of smoke evacuation systems to safeguard surgical personnel against the harmful effects of surgical smoke, which contains hazardous substances such as viruses, bacteria, and carcinogens.
In response to these laws, many healthcare facilities are investing in smoke evacuation systems tailored for laparoscopic procedures, resulting in a rise in demand for insufflators compatible with such systems. Moreover, certain insufflator manufacturers have developed insufflators with integrated smoke evacuation capabilities, enabling healthcare facilities to comply with these laws while reducing the need for extra equipment and tubing. Overall, the implementation of surgical smoke evacuation laws has had a positive impact on the laparoscopic insufflator market, with an increased demand for compatible systems driving manufacturers to create new technologies.
There are currently eleven states with smoke evacuation laws and ten currently pending legislation as of 2023 (AORN, 2023). The resultant growth in segments related to smoke evacuation, such as the insufflation tubing market, can be attributed to this trend. Healthcare facilities located in states without such legislation are also opting to purchase insufflators with smoke evacuation capabilities due to the documented risks associated with surgical smoke (AORN, 2022). The number of states mandating smoke evacuation systems is expected to rise over the forecast period, leading to continued growth in Insufflation market.
Chart 1: Insufflation Device Market Value by Segment, United States, 2019-2029
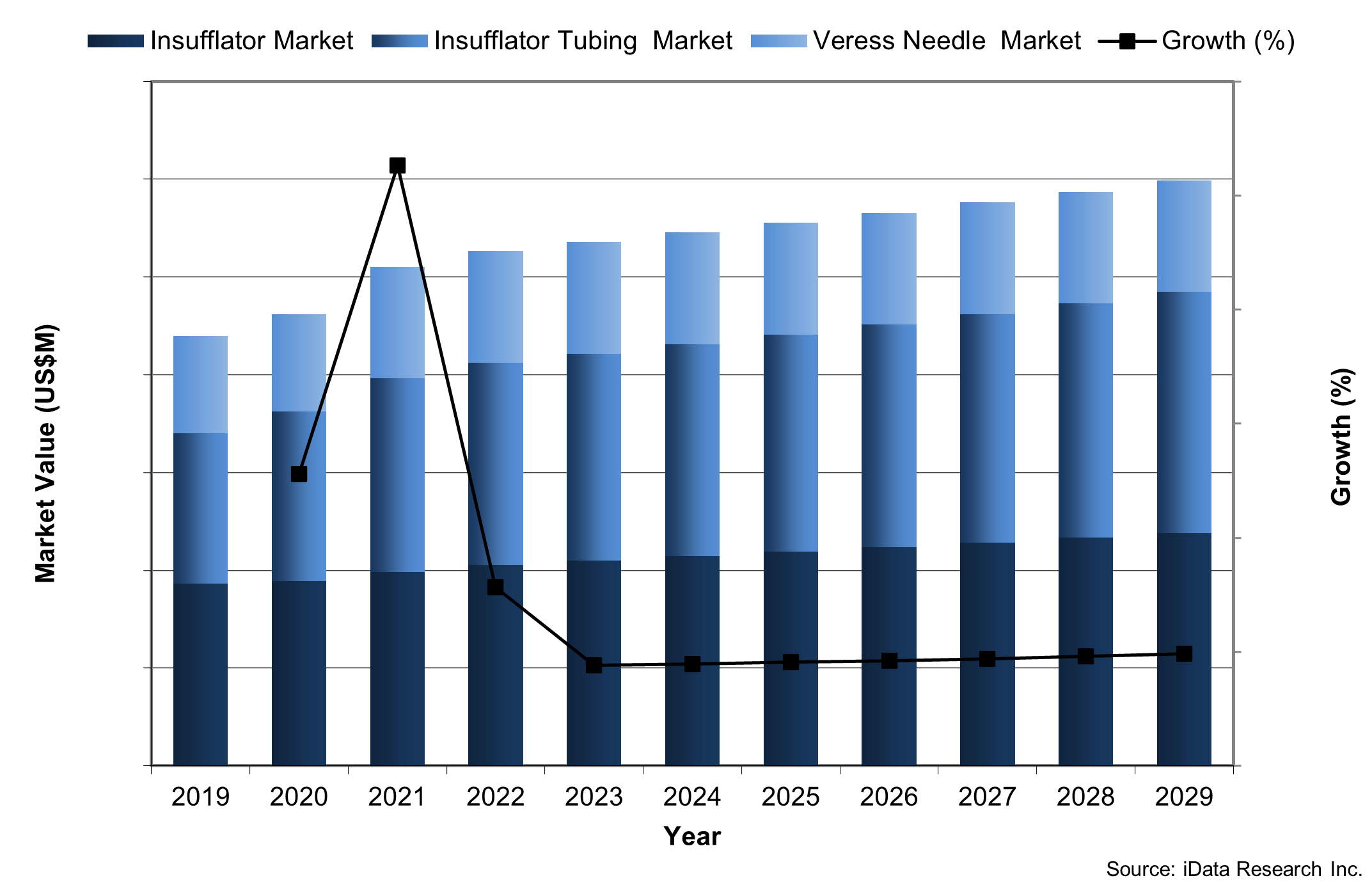
Powered Morcellation
In recent years, the safety concerns about power morcellators in gynecological surgery have significantly impacted the laparoscopic morcellation market in the US. Power morcellators break down large masses of tissue in laparoscopic surgery, but the FDA recommends that they be used only with a tissue containment system that can remove potentially cancerous tissue (FDA, 2022) and in select patients (FDA, 2020).
Due to FDA warnings and restrictions from healthcare facilities and insurance companies, the use of power morcellators has decreased. Consequently, some manufacturers have shifted their focus to other areas of laparoscopic surgery, such as suturing and stapling devices, while others have developed alternative technologies for tissue removal.
The current state of the US laparoscopic morcellation market is marked by a decline in demand for power morcellators and an increase in interest in alternative technologies. The market is expected to continue evolving in response to safety concerns and new technological developments.
Direct Energy Devices
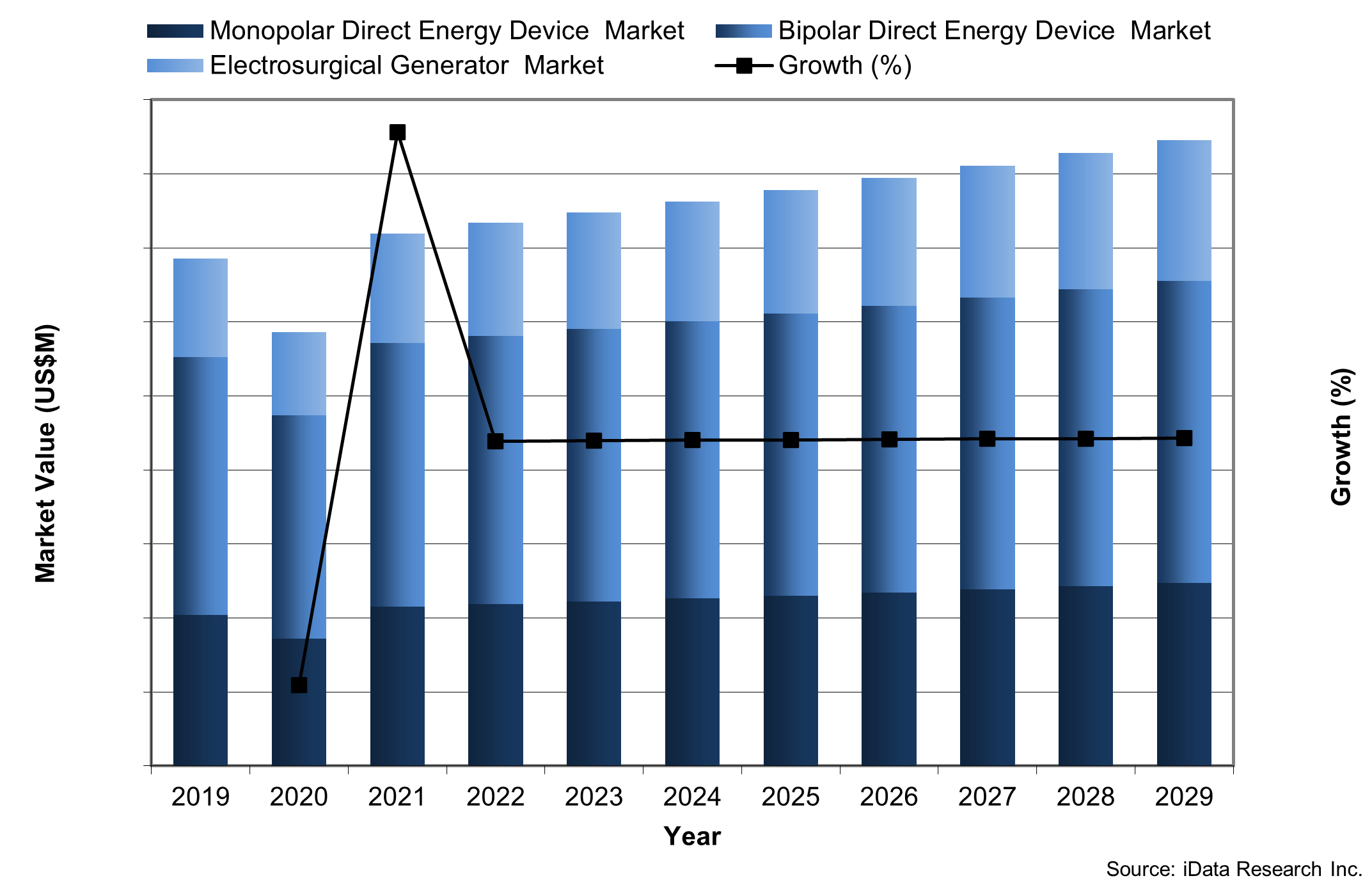
The laparoscopic direct energy device market in the US is growing due to technological advancements and the rise of minimally invasive surgery. Direct energy devices, such as electrosurgical instruments and ultrasonic devices, are used to cut, coagulate and seal tissue during laparoscopic procedures.
The market is experiencing trends in the development of advanced direct energy devices that provide greater precision, safety, and efficiency, as well as specialized energy devices tailored to specific surgical specialties. However, challenges such as the high cost of advanced devices and the need for specialized surgeon training and education remain. There are also concerns about the potential risks of thermal injury and electrical burns during laparoscopic procedures.
The direct energy device market is in a healthy position, with ongoing innovation driving growth and expanding applications. Nevertheless, it is critical for manufacturers and healthcare providers to balance the benefits of direct energy devices with the potential risks and ensure appropriate training and safety measures are in place to minimize complications.
Chart 2: Direct Energy Device Market Value by Segment, United States, 2019-2029
Disposable vs Reusable
The COVID-19 pandemic has had a significant impact on the use of laparoscopic devices in the US, leading to a shift in trends towards using disposable over reusable devices. Due to concerns about potential transmission of the virus via reusable devices, healthcare facilities initially increased their use of disposable devices such as trocars, graspers and scissors. Manufacturers responded to the increased demand by expanding their product lines and developing new disposable devices.
However, the environmental impact of the increased use of disposable devices is also a concern, prompting a renewed focus on reusables for high-cost and high-volume devices like laparoscopic staplers and energy devices (Freund, J, et al., 2022; Maurice, J. et al., 2020). This trend has led to the development of more cost-effective and easier to maintain reusable devices. The US laparoscopic device market is still adapting to these changes, as healthcare facilities and manufacturers aim to balance safety, convenience and cost-effectiveness.
Conclusion
The US laparoscopy market is growing fast due to a rising demand for minimally invasive surgeries and advancements in technology. New laws regarding surgical smoke evacuation have boosted the insufflation market, while safety concerns over power morcellators have led to interest in alternative technologies. The direct energy device market is developing advanced devices that provide greater precision, safety, and efficiency, but cost and specialized surgeon training are challenges. The industry offers many opportunities for innovation and advancement, but it is important to understand trends and challenges to anticipate their impact on the market.
For further insights, please see the report Laparoscopic Market Size, Share & COVID-19 Impact Analysis - United States - 2023-2029 by iData Research.
About the Authors
Nathaniel Bool is a research analyst at iData Research. He develops and composes syndicated research projects regarding the medical device industry, publishing the U.S. Laparoscopic device report series.
Kamran Zamanian, Ph.D., is CEO and founding partner of iData Research. He has spent over 20 years working in the market research industry with a dedication to the study of medical devices used in the health of patients all over the globe.
About iData Research
For 16 years, iData Research has been a strong advocate for data-driven decision-making within the global medical device, dental and pharmaceutical industries. By providing custom research and consulting solutions, iData empowers its clients to trust the source of data and make important strategic decisions with confidence.
Works Cited
FDA. (2020). Update: Perform Only Contained Morcellation When LPM Is Appropriate. U.S. Food and Drug Administration.
FDA. (2022). Laparoscopic Power Morcellator. U.S. Food and Drug Administration.
Freund, J., Gast, K., Zuegge, K., & Hicks, A. (2022). Environmental considerations in the selection of medical staplers: A comparative life cycle assessment. Journal of Cleaner Production, 371, 133490.
Maurice, J. B., Siau, K., Sebastian, S., Ahuja, N., Wesley, E., Stableforth, W., & Hayee, B. (2020). Green Endoscopy: A Call for Sustainability in the Midst of Covid-19. The Lancet Gastroenterology & Hepatology, 5(7), 636–638.
McDonald, N. H. (2022, November 23). Fighting for Smoke-Free ORs. Outpatient Surgery Magazine.
National Map of Current Surgical Smoke Evacuation Laws and Legislation. Aorn.org. (n.d.).

